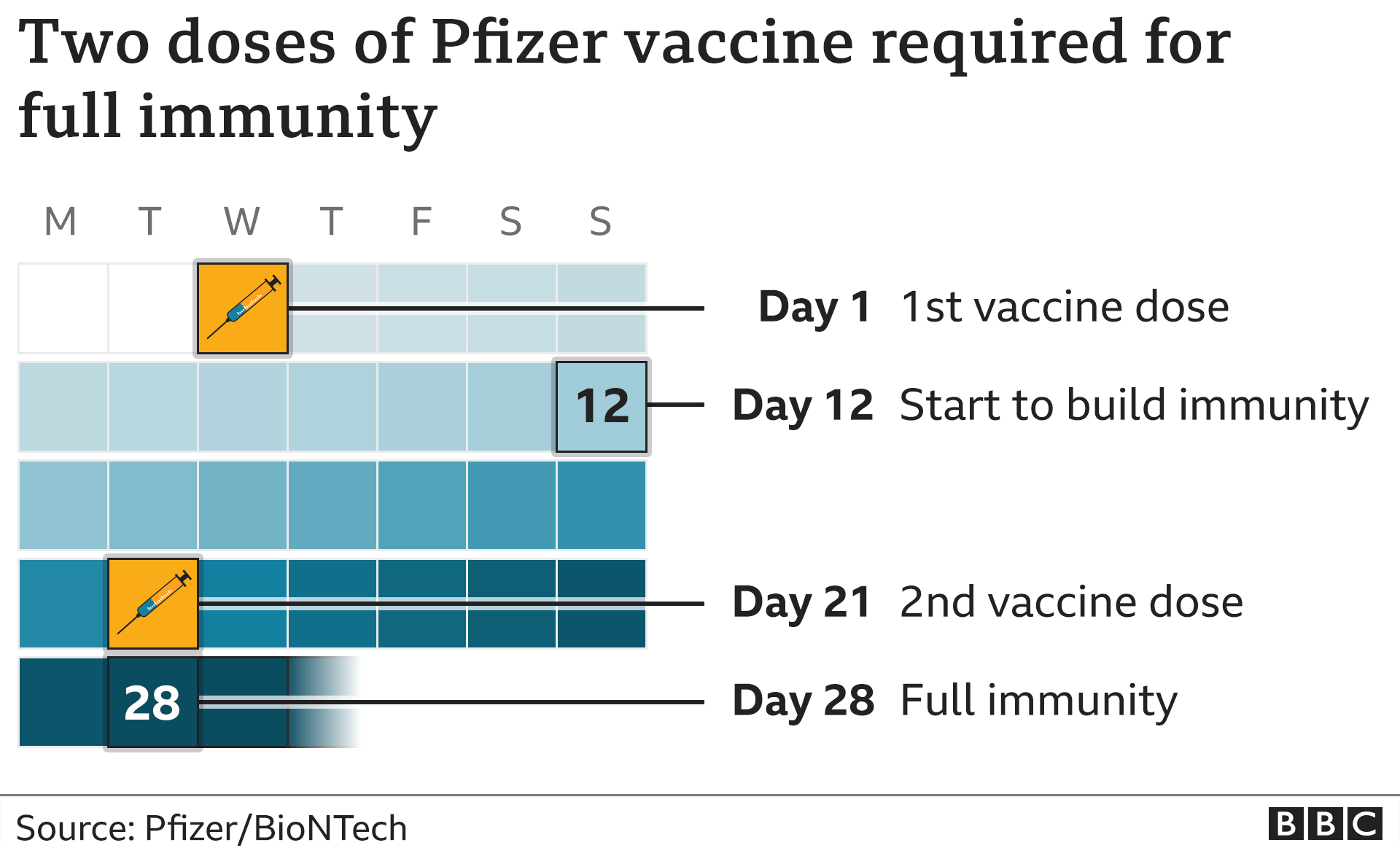
The vaccine is not approved for people under age 18 in the United States. vaccine safety monitoring databases has also suggested that Moderna's vaccine may carry a higher risk of myocarditis among young people. The benefits of shots based on so-called mRNA technology used by both Moderna and Pfizer-BioNTech in preventing COVID-19 continue to outweigh the risks, regulators in the United States, EU and the World Health Organization have said.ĭata suggests reported cases of rare heart inflammation are relatively higher after Moderna's vaccine compared with the Pfizer/BioNTech shots, Canadian health officials said last week.Īlthough both vaccines are based on mRNA technology, the Pfizer shot contains 30 micrograms of vaccine per dose compared with 100 micrograms in the Moderna vaccine.ĭata from one of two U.S. The EMA's safety committee concluded in July that inflammatory heart conditions can occur in very rare cases following vaccination with Comirnaty or Spikevax, more often in younger men after the second dose. The Danish Health Authority said it had made the decision even as "heart inflammation is an extremely rare side effect that often has a mild course and goes away on its own". REUTERS/Juan Medina Acquire Licensing Rights Learn more about COVID-19 vaccines and specific information for people who are moderately or severely immunocompromised.A nurse prepares a syringe with a dose of the Moderna coronavirus disease (COVID-19) vaccine at Enfermera Isabel Zendal hospital in Madrid, Spain, July 23, 2021. Review vaccination benefits, risks, scientific evidence, and additional resources, in our public health recommendation. People speak with their health care provider to evaluate the risks and benefits of vaccination for them.Vaccination especially for those 60 and older, and people with other medical conditions.Vaccination for all eligible people 6 months of age and older.As always, DHEC also recommends and encourages people to speak with their health care provider to evaluate the risks and benefits of vaccination. This recommendation extends especially to those 60 and older, and those with other medical conditions that may cause complications from COVID-19 infection. 
Although the vaccine may not provide 100% protection against infection, it is likely to prevent the most harmful outcomes of COVID-19. Having reviewed the available data and side effect profiles for the updated COVID-19 vaccine (2023-2024), DHEC recommends that all eligible people 6 months of age and older receive this important, updated vaccine. 12, 2023 DHEC Recommends Updated COVID-19 Vaccination Bureau of Environmental Health Services.All Forms, Applications, Registrations, Reporting.FORMS: Applications, Registration, Reporting, Etc.Applying for a Permit, License, Certification or Registration from DHEC.Hospital-Based Crisis Stabilization Units.Emergency Medical Technician (EMT) Certification Requirements.Health Facility Regulations, Licensing, CON.Residential Treatment Facilities for Children & Adolescents.Health Facilities: How to File a Complaint.New Schedules for Controlled Substances.Changes - Name, Address, Drug Schedules.Environmental Public Health Fact Sheets.West Ashley High School TB Investigation.Child & Adolescent Immunizations and Vaccines.Children's Rehabilitative Services (CRS).Women, Infants, & Nutrition Program (WIC).State Statistics on Family Planning & Pregnancy.Manufactured Food Safety in South Carolina.Regulation - Small Business Assistance Program.Watershed-Based Plan Development Grants.Beachfront Jurisdictional Lines Stakeholder Workgroup.Oil & Natural Gas and Terminal Facility Registration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
